Low density, high strength and high hardness are characteristics shared with many other ceramics. Read More…
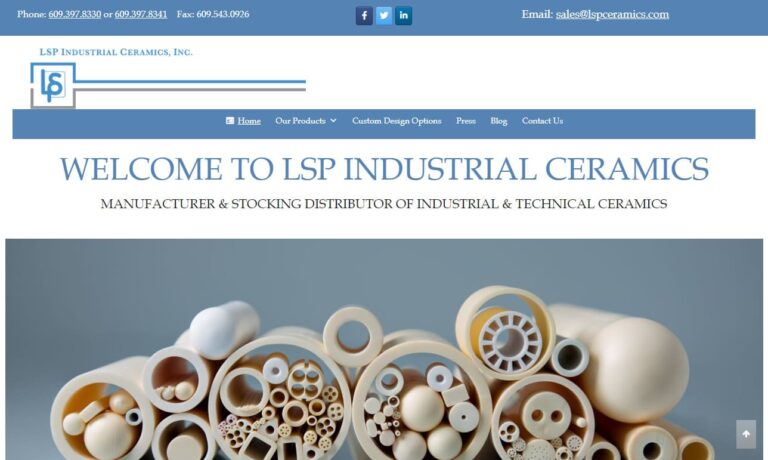
As a manufacturer and stocking distributor of industrial and technical ceramics, LSP carries the most diversified inventory of ceramic tubes, spacers, bushings, etc. in the industry.
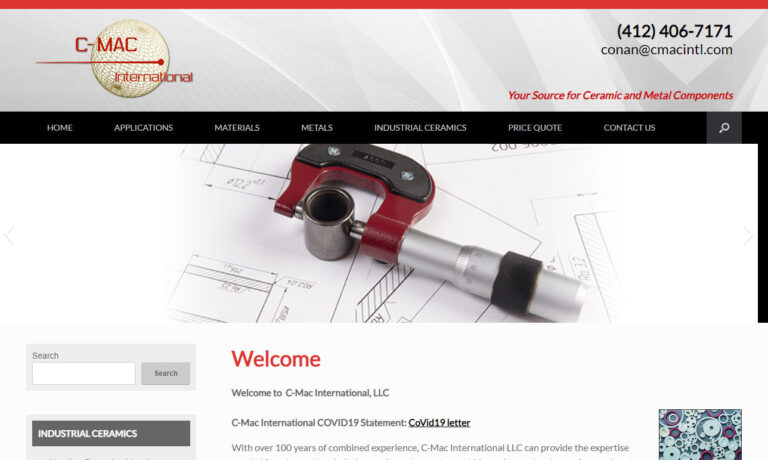
C-Mac International manufactures custom advanced technical ceramic solutions. Our specialties are Zirconia (MgO stabilized and Yttria stabilized), Alumina (90%, 96%, and 99.5% purity), and Tungsten Carbide (Cobalt and Nickel Binder). We also work with steatite, cordierite, silicon nitride, ceramic crucibles, and crushable ceramics. We prioritize customer needs - we have a 48-hour delivery on...
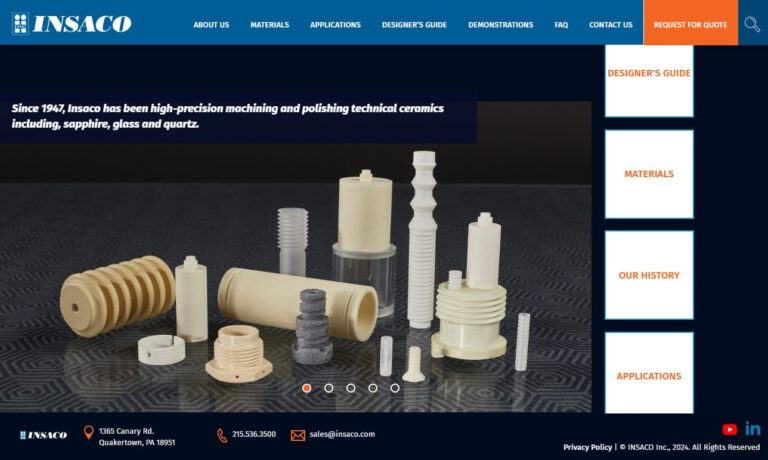
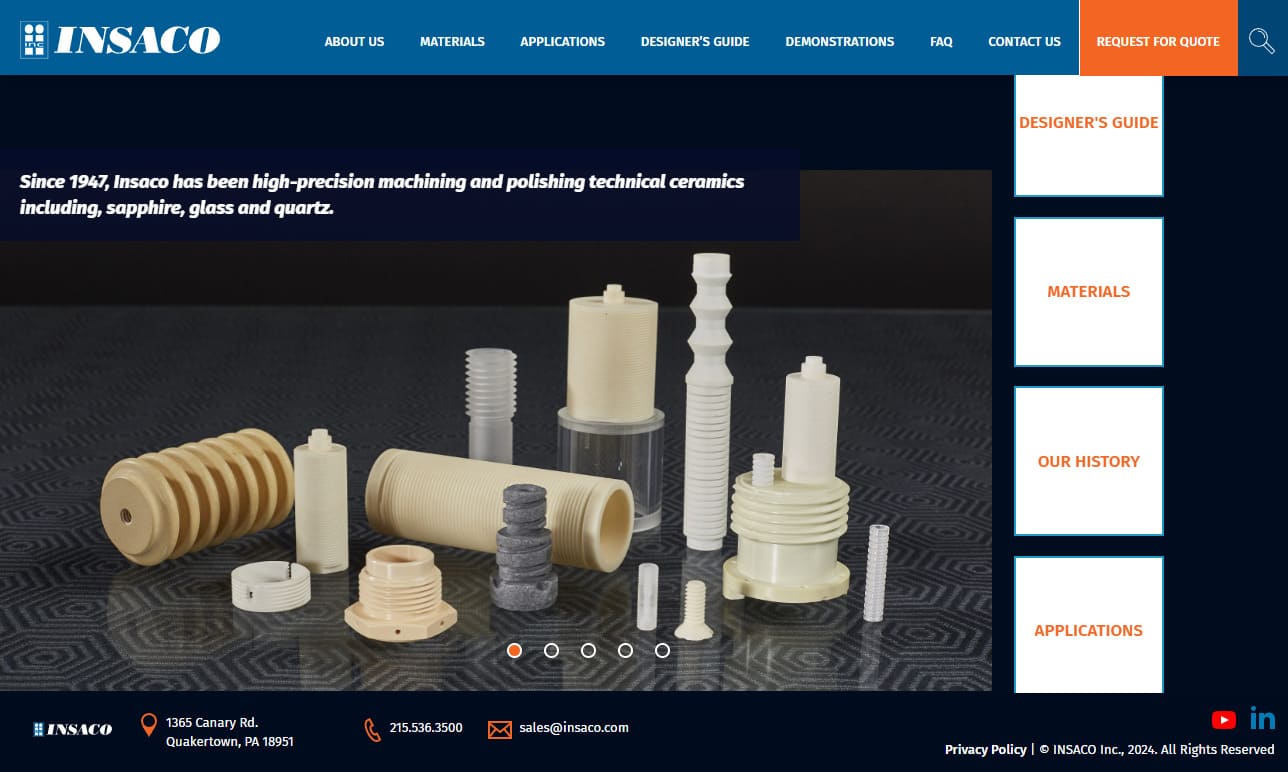
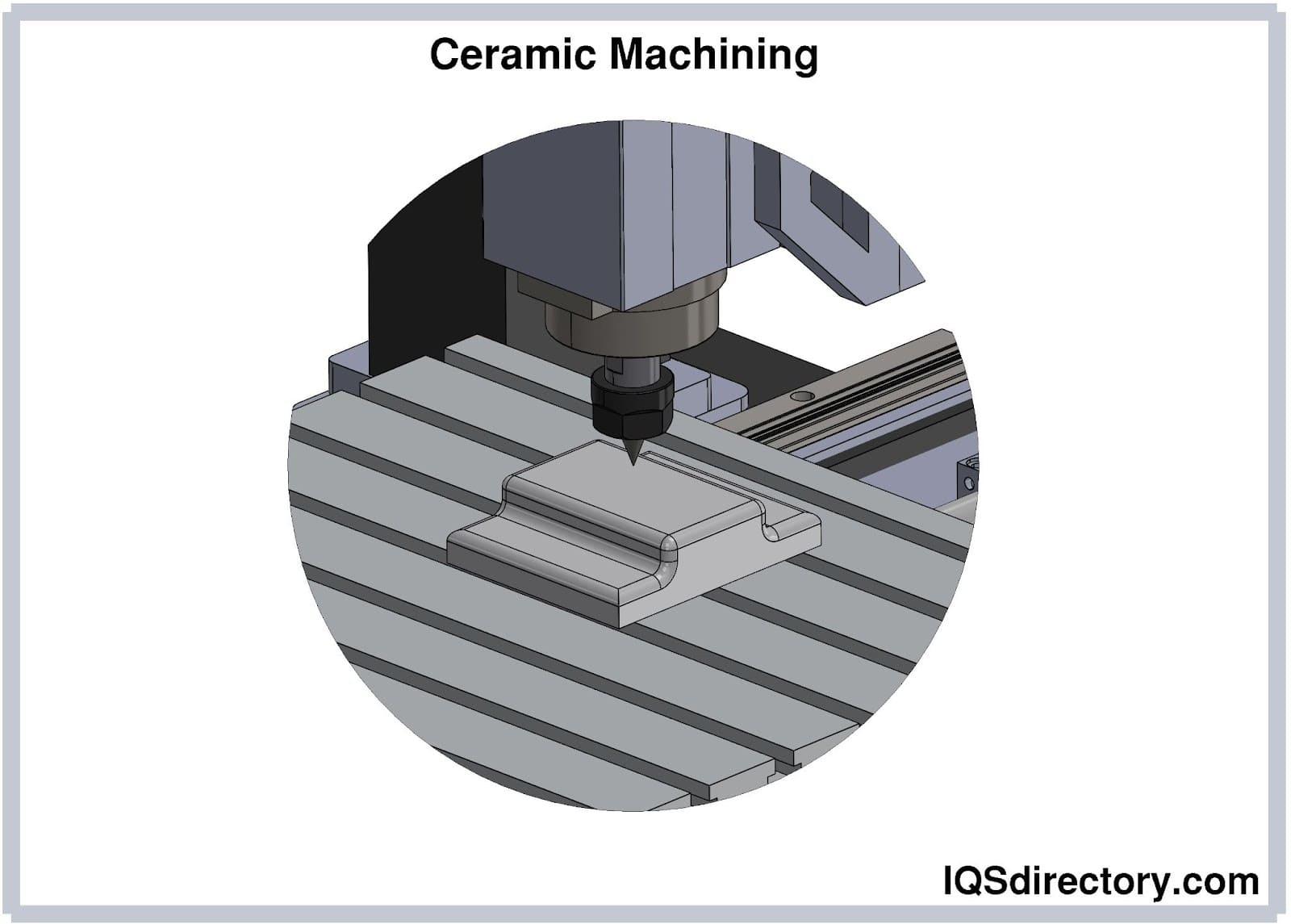
Insaco provides custom grinding and machining services to fabricate precision parts from sapphire, quartz, and most technical ceramics including alumina, zirconia, silicon carbide, silicon nitride, aluminum nitride, and others.
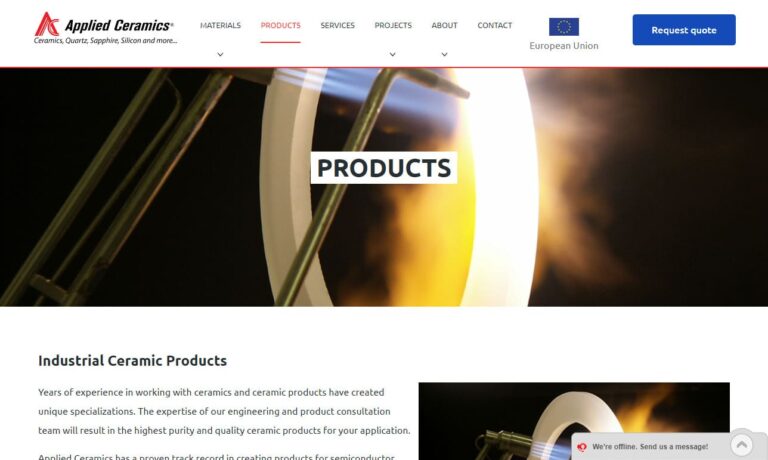
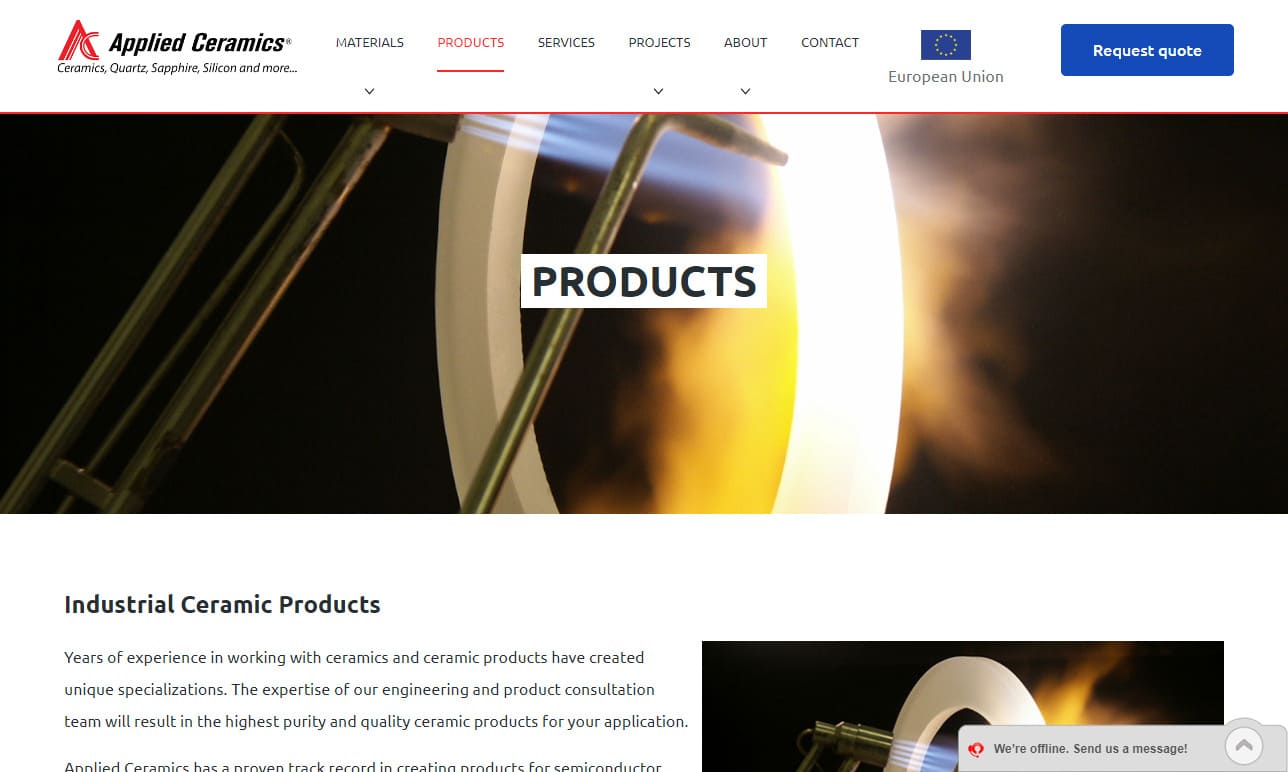
Applied Ceramics is a fabricator of custom-made ceramic parts designed for semiconductor, solar, fuel cell, oil drilling, nuclear, and numerous other industries. Materials include ACI-995 Alumina, Zirconia, and more. Our extensive experience with precision designs supported by our team of specialists ensures that our customers have the ideal solution to meet the needs of their application. To get ...
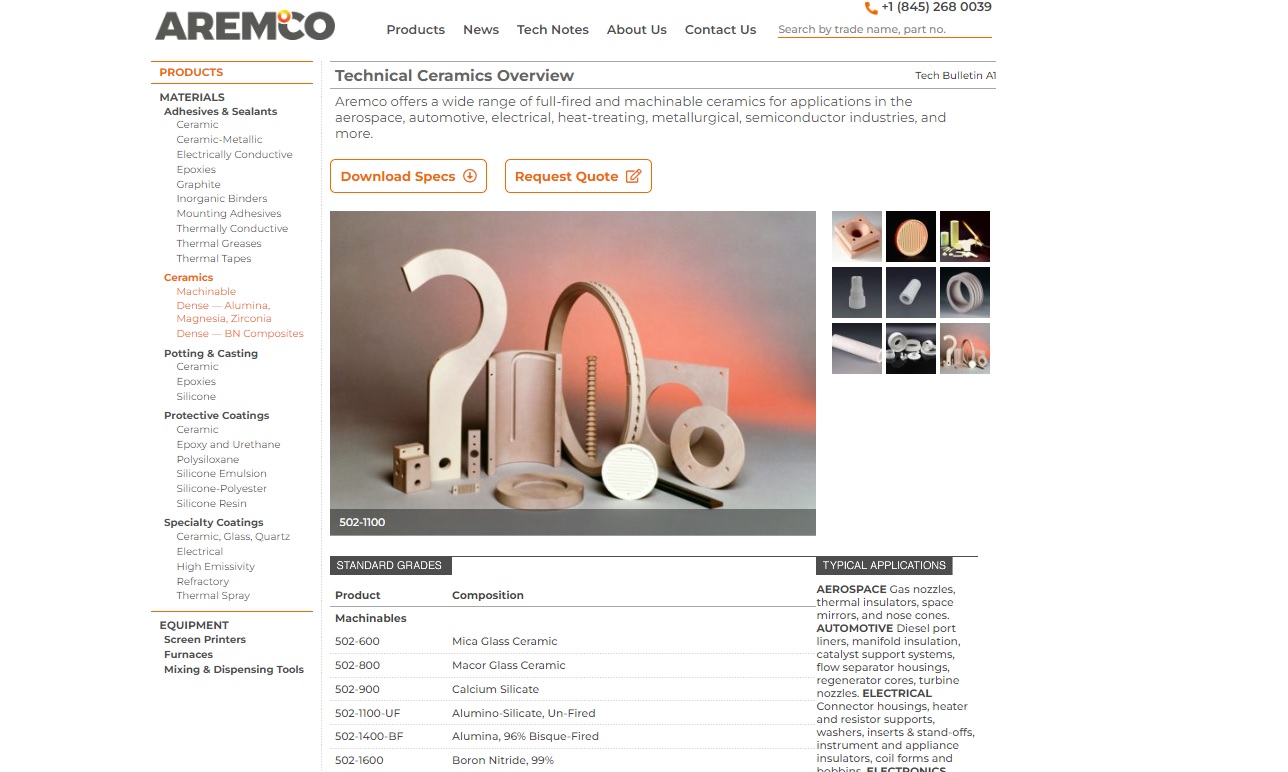
Aremco is a leader in the custom formulation of advanced industrial materials including technical ceramics. Offering many capabilities for a broad range of machinable & dense ceramic materials, Aremco serves aerospace, automotive, electrical, electronics, heat treating, metallurgical, petrochemical & plastics applications with superior finished ceramic parts. 100’s of standard industrial...

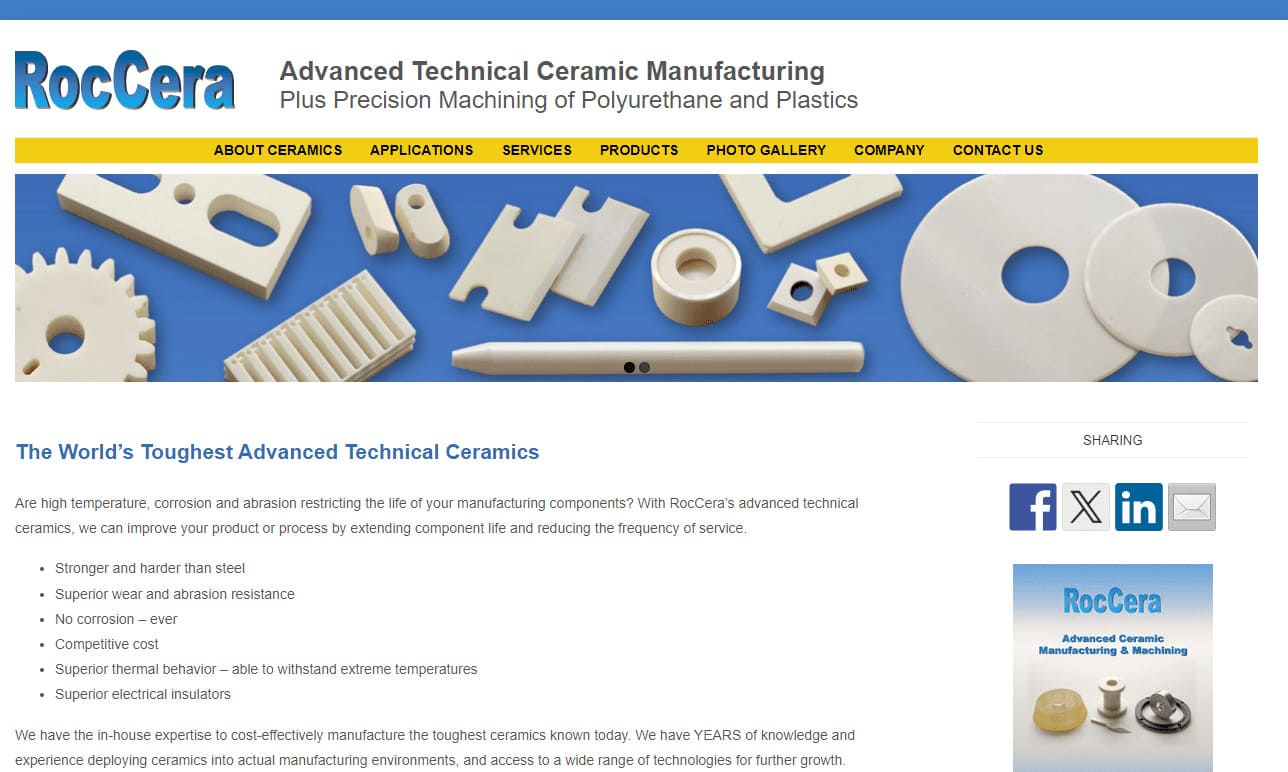
RocCera designs high-performance ceramic components. With a relentless pursuit of excellence and innovation, we have positioned ourselves as a leading provider of custom ceramic solutions catering to a diverse range of industries. Specializing in advanced technical ceramics, RocCera boasts a versatile portfolio that includes the manufacturing of ceramic components for various applications. Our...
As an ISO 9000 and QS 9000 company, CeramTec North America offers ceramic products such as ceramic insulators, ceramic cutting tools, ceramic substrates and ceramic-to-metal joints. We are one of the world's largest ceramic manufacturers that offer custom-engineered high-tech ceramics.
More Silicon Carbide Manufacturers
Silicon carbide offers low thermal expansion, high thermal conductivity, strong thermal shock resistance and elevated chemical intertness as it is not attacked by acids, alkalis or molten salts. A modern ceramics material, silicon carbide is classified as an advanced, technical, or engineering ceramic due to its superior attributes. Silicon carbide components are common to industrial environments as they are particularly well suited to the high stress needs of these settings.
Electrical conduction, semiconductors, automotive, structural and abrasion industries are just a few of the many sectors in which silicon carbide is utilized. Grinding machines, brake discs, seals, bearings and heat exchangers are all commonly made of this material, maximizing its heat resistance and conductivity. Silicon carbide grit can also be used to create ground glass for use in photographic equipment and in the creation of decorative glassware.
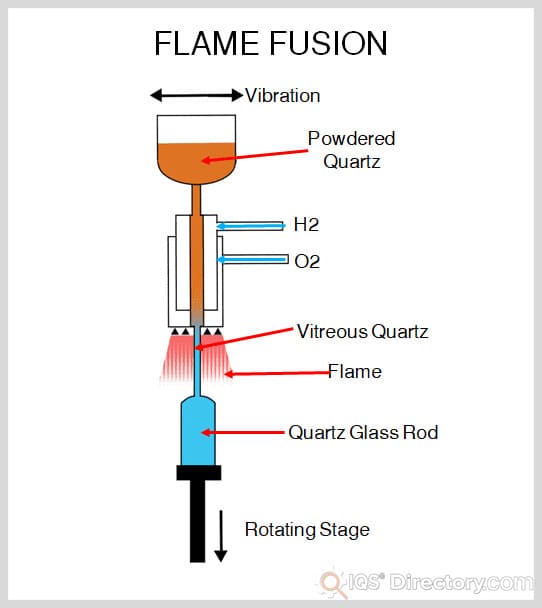
Although natural occurrence of silicon carbide is rare, several processes allow for large production of man-made replicas of this substance. The Ascheson furnace and Lely are commonly used methods. The Ascheon technique involves heating silica, carbon and other additives that increase porosity to maximum temperatures and then gradually lowering them. The Lely process sublimates the particles in an argon atmosphere by heating them in a fashion similar to the Ascheson furnace. Chemical vapor deposition is sometimes used to produce cubic silicon carbide, though it is a very expensive option.
Thermal decomposition of polymethylsilyne also yields pure silicon carbide when it occurs in an inert atmosphere with low temperatures. This process has the added advantage of pre-forming the polymer before it is transformed into a ceramic. When silicon carbide is formed, no matter the process, the color of the crystals helps to determine the purity of a given sample. Colorless, pale yellow and green crystals are the purest. Brown, blue or black crystals indicate some degree of contamination and are less pure.
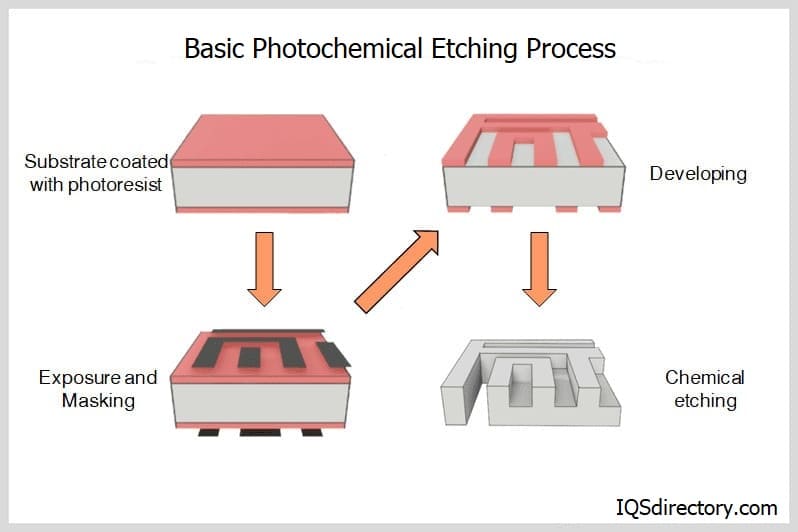
Common imputiries include nitrogen, aluminum and iron, all of which affect the electrical conductivity of the final product. The crystals or grains produced by any of these methods are then bonded together by manufacturing techniques such as sintering, firing, hot pressing, hipping, extrusion, fusing, slip or pressure casting, injection molding and deposition.







 Adhesives
Adhesives Alumina Ceramic
Alumina Ceramic Ceramic
Ceramic Glass
Glass Graphite
Graphite Lubricants
Lubricants Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services