Alumina crucibles are specific kinds constructed from aluminum oxide, also known as alumina, the same raw material used to make aluminum metal. Crucibles are built of high-temperature resistant materials and used in chemistry labs as containers for extremely hot chemical substances. In addition, alumina is frequently utilized in ceramic form due to its strength, inexpensive cost, and capacity to tolerate temperatures of up to 3,272 °F (1,800 °C). Read More…
As a manufacturer and stocking distributor of industrial and technical ceramics, LSP carries the most diversified inventory of ceramic tubes, spacers, bushings, etc. in the industry.
C-Mac International manufactures custom advanced technical ceramic solutions. Our specialties are Zirconia (MgO stabilized and Yttria stabilized), Alumina (90%, 96%, and 99.5% purity), and Tungsten Carbide (Cobalt and Nickel Binder). We also work with steatite, cordierite, silicon nitride, ceramic crucibles, and crushable ceramics. We prioritize customer needs - we have a 48-hour delivery on...
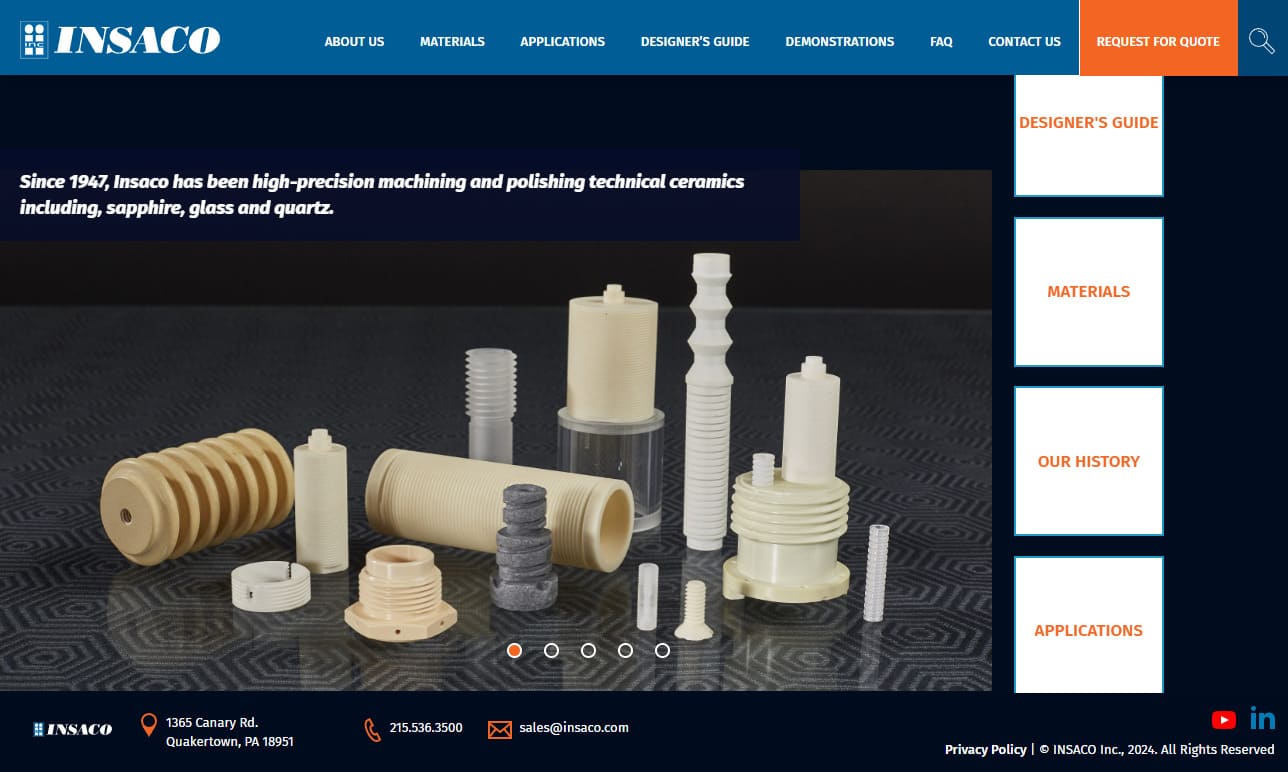
Insaco provides custom grinding and machining services to fabricate precision parts from sapphire, quartz, and most technical ceramics including alumina, zirconia, silicon carbide, silicon nitride, aluminum nitride, and others.
Applied Ceramics is a fabricator of custom-made ceramic parts designed for semiconductor, solar, fuel cell, oil drilling, nuclear, and numerous other industries. Materials include ACI-995 Alumina, Zirconia, and more. Our extensive experience with precision designs supported by our team of specialists ensures that our customers have the ideal solution to meet the needs of their application. To get ...
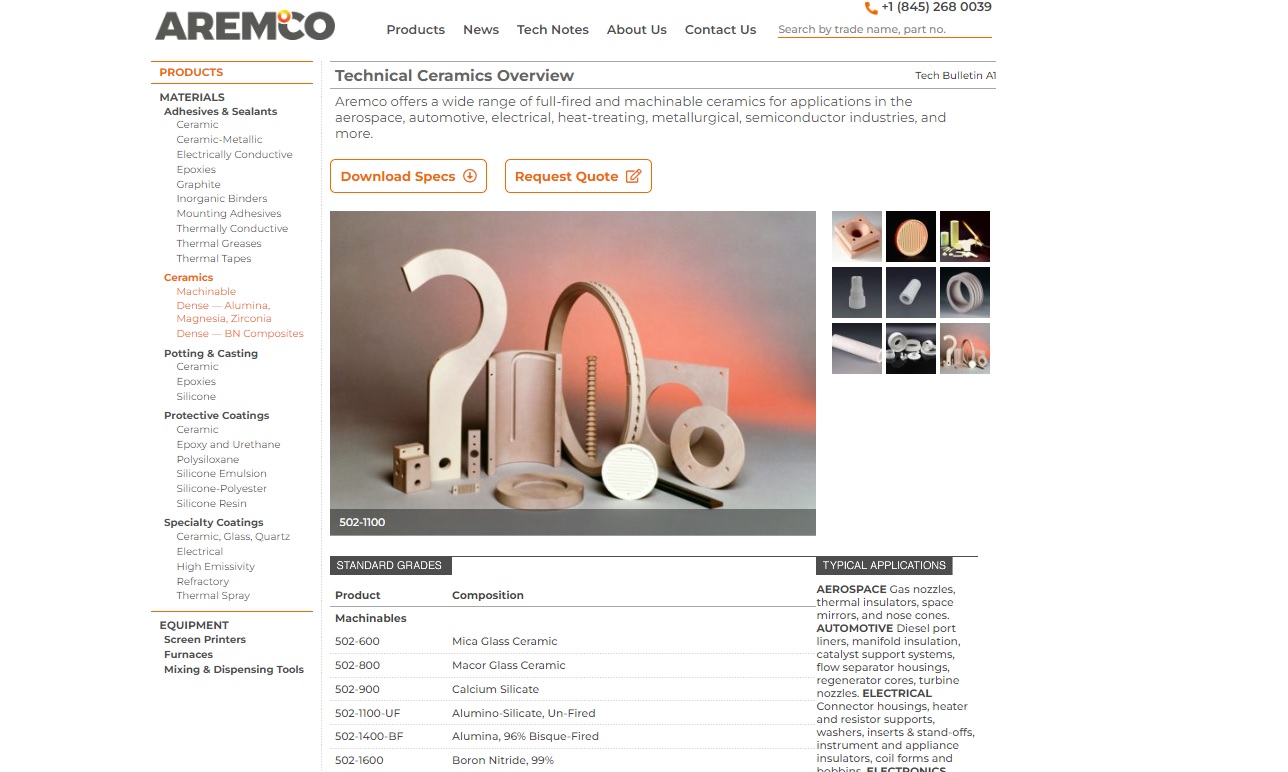
Aremco is a leader in the custom formulation of advanced industrial materials including technical ceramics. Offering many capabilities for a broad range of machinable & dense ceramic materials, Aremco serves aerospace, automotive, electrical, electronics, heat treating, metallurgical, petrochemical & plastics applications with superior finished ceramic parts. 100’s of standard industrial...

RocCera designs high-performance ceramic components. With a relentless pursuit of excellence and innovation, we have positioned ourselves as a leading provider of custom ceramic solutions catering to a diverse range of industries. Specializing in advanced technical ceramics, RocCera boasts a versatile portfolio that includes the manufacturing of ceramic components for various applications. Our...
As an ISO 9000 and QS 9000 company, CeramTec North America offers ceramic products such as ceramic insulators, ceramic cutting tools, ceramic substrates and ceramic-to-metal joints. We are one of the world's largest ceramic manufacturers that offer custom-engineered high-tech ceramics.
More Alumina Crucible Manufacturers
Laboratory research, academia, and industry all use alumina crucibles, which come in various shapes and sizes. Due to its adaptability and affordable price as a material, alumina has many uses. Alumina is a good material to survive high temperatures and chemical corrosion because it has a high melting point, strong hardness, and good chemical stability. There are numerous sizes and types of alumina crucibles available.

How Crucibles are Made
Bauxite ore, which includes aluminum, is treated industrially to produce aluminum oxide. Calcined alumina, which predominantly comprises alumina with certain impurities, mostly sodium oxide, results from numerous purifying procedures. As a result, crucibles are frequently created using calcined alumina. Similar techniques can also produce high-purity alumina, frequently favored in industrial laboratories due to its superior resilience to extremely high temperatures.
Materials Used to Produce Alumina Crucibles
The ability to sustain extremely high temperatures without degrading makes pure alumina a refractory substance; 3,760°F is roughly the temperature at which aluminum melts. Crucibles are typically constructed using aluminum oxide, a kind of alumina. From the aluminum-containing mineral bauxite, aluminum oxide is industrially processed.
Calcined alumina is obtained after several purification procedures; it mostly includes alumina with certain impurities, primarily sodium oxide. Alumina's heat resistance decreases, unfortunately, with decreasing purity. High-purity alumina is favored because of this, especially in industrial laboratories. Similar to calcined alumina, this alumina can be produced by the same method.
Applications of Alumina Crucibles
Due to its usefulness in chemical laboratories as containers for highly hot chemical compounds, alumina crucibles' heat tolerance is a particularly important quality. There are numerous uses for alumina crucibles in education, industry, and laboratory research. For instance, high-form alumina crucibles are frequently utilized in academic labs. These "crucibles," resemble a deep, narrow cup, are frequently used to melt or heat chemicals in an oven or over a Bunsen burner. High-form crucibles are offered in rectangular and cylindrical shapes commercially.
Other crucibles are typically small, have a diameter smaller than an adult human hand, and are suitable for holding insufficient chemicals needed in academic or research labs. All smaller crucibles are used with a lid made of the same material and are handled with tongs to avoid burning or other mishaps. Industrial alumina crucibles, which can be used to melt metals and make alloys, can be much larger and need to be installed in the furnace with enough room and outside support.

Considerations When Choosing a Crucible
When choosing a crucible, there are several aspects to take into account. As a result, the task may appear overwhelming. These variables can include the required volume, the form, and the chemical reactions between the chemicals placed in the crucible and the crucible itself, to name a few. Here is a list of some of the most crucial things to consider while choosing a crucible.
- The degree of heat the crucible must withstand
- The type of crucible material that will be chosen
- The ramp rate must not cause the crucible to undergo thermal shock
- The crucible must have a heat gradient
- The size of the crucible required
- Considering if the crucible's geometry is significant
- Fitting of the crucible into the furnace
- Can the crucible be used a second time?
Advantages of Alumina Crucibles
- Alumina crucibles are simple to clean and maintain. Bright and delicate, the glazed surface of the alumina ceramic crucible is simple to wipe away after becoming polluted.
- Porcelain has a poor water absorption rate and very few pores. To store the solution, use an alumina ceramic crucible. Once the mouth is tightly closed, the solution can be protected from external microorganisms, volatilization, and penetration.
- Alumina provides durable and stable chemical characteristics. Alumina is superior to things made of metals like copper, iron, aluminum, etc. For example, crucibles made of alumina ceramic have some resistance to acid, alkali, salt, and atmospheric carbon dioxide. They are also not easily chemically reacted with these substances and do not rust.
- Alumina has stable thermal response and sluggish heat transfer. The alumina ceramic crucible has the property of not easily bursting when subjected to a certain temperature differential and swift changes in cold and heat. Glassware is inferior to this. Heat transfer is sluggish because it is a poor heat conductor. As a result, hands won't be burned when holding it to hold hot liquids like boiling water.

Choosing the Correct Alumina Crucibles Manufacturer
To make sure you have the most constructive outcome when purchasing Alumina Crucibles from an Alumina Crucibles Supplier, it is important to compare at least 5 Companies using our list of Alumina Crucibles companies. Each Alumina Crucibles Manufacturer has a business profile page that highlights their areas of experience and capabilities and a contact form to directly communicate with the manufacturer for more information or request a quote. Review each Alumina Crucibles business website using our patented website previewer to get an idea of what each company specializes in, and then use our simple RFQ form to contact multiple Alumina Crucibles companies with the same quote.







 Adhesives
Adhesives Alumina Ceramic
Alumina Ceramic Ceramic
Ceramic Glass
Glass Graphite
Graphite Lubricants
Lubricants Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services